BioElectronics Clinical Studies: Postoperative Pain & Edema
Postoperative Pain
Pulsed Electromagnetic Fields for Postsurgical Pain Management in Women Undergoing Cesarean Section: A Randomized, Double-Blind, Placebo-controlled Trial
Khooshideh M, Latifi Rostami SS, Sheikh M, Ghorbani Yekta B, Shahriari A.
Objectives: To evaluate the efficacy of pulsed electromagnetic field
(PEMF) in relation to reducing postoperative pain, analgesic use,
and wound healing in patients undergoing Cesarean section
(C-section).
Methods: This randomized, double-blind, placebo-controlled trial
evaluated 72 women who underwent elective C-section. Thirty-six
patients were assigned to the active-PEMF and 36 to the sham-
PEMF groups. The participants were asked to report their pain
intensity on a Visual Analog Scale (VAS) at 2, 4, 6, 12, and 24
hours and 2, 4, and 7 days after surgery. The amount of analgesics
used was recorded. The surgical site was evaluated to assess the
wound-healing process on the seventh postoperative day.
Results: Postoperative pain VAS scores were significantly lower in
the active-PEMF group in all the measured periods within the early
and the late postoperative periods. Fewer women in the active-
PEMF group experienced severe postoperative pain within 24
hours postoperatively (36% vs. 72%, P=0.002). Analgesic use
during the first 24 hours after C-section was 1.9-times lower in the
active-PEMF group (1.6±0.7 vs. 3.1±1.2, P<0.001). The total
analgesic use during the seventh postoperative days was 2.1-times
lower in the active-PEMF group than in the sham group (1.7±0.7
vs. 3.7±1.1, P<0.001). Seven days postoperatively, patients in
the active-PEMF group had better wound healing with no exudate,
erythema, or edema (P=0.02).
See Complete C-Section Study here
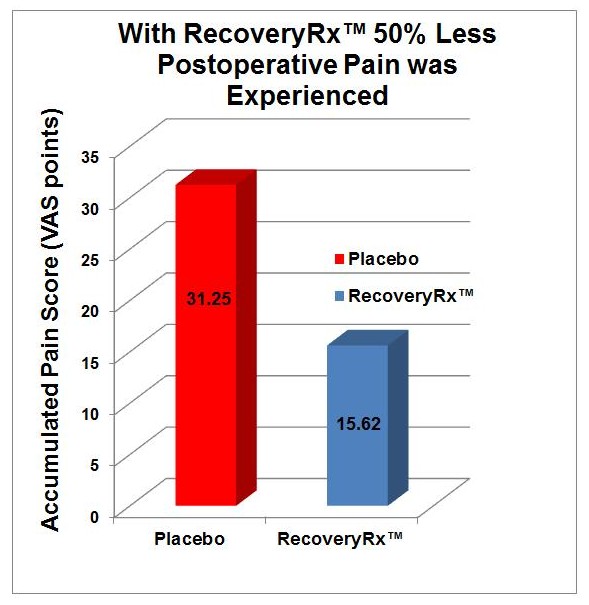
Control of Postoperative Pain with a Wearable Continuously Operating Pulsed Radiofrequency Energy Device: A Preliminary Study
Ian M. Rawe, Adam Lowenstein, C. Raul Barcelo & David G. Genecov
BioElectronics RecoveryRx® was investigated in a double blind, randomized and placebo controlled study for postoperative pain and narcotic pain medication use following breast augmentation surgery. Data, VAS pain scores (0-10) and medication was collected for seven days post-surgery. The group using RecoveryRx® experienced 50% less pain and consumed significantly less narcotic pain medications. The study is published in the Aesthetics of Plastic Surgery.
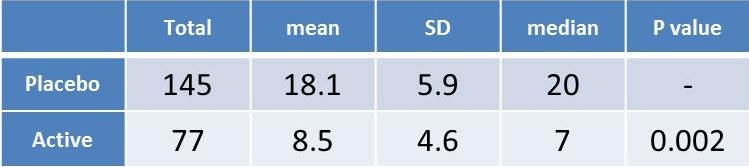
Table 1. Narcotic pain medication use (total pills) was significantly lower in the group using RecoveryRx®
View Complete Postoperative Clinical Studies
Postoperative Pain & Edema
Accelerated recovery of post-operative dental implant patients by means of pulsed shortwave (SWT) therapy: An Observational Study
Operti & Tealdo March 2014
The purpose of this white paper is to report on the experience and on the clinical outcome resulting from the use of a wearable electromagnetic device (RecoveryRx Therapy by BioElectronics Corp., Frederick, MD USA) in post-operative implant patients and to evaluate the efficacy in reducing edema, inflammation and pain immediately after dental implant surgery. The device is known to promote faster than normal healing process, anticipating masticatory function.
View Complete Observational White Paper
Postoperative Edema
Use of Radio-Frequency Pulsed Energy in the Control of Postoperative Reaction in Blepharoplasty
Frederick V. Nicolle, M. Chir., F.R.C.S. and Richard M. Bentall, F.R.C.S.
Development of BioElectronics medical devices was based on the pioneering basic and clinical research by Dr Richard Bentall. In an initial study Dr. Bentall looked at the effect of radiofrequency electromagnetic fields on postoperative edema from blepharoplasty (eyelid surgery). This pioneering study was published in the Aesthetics of Plastic Surgery.