The Opioid Epidemic is Fueled by the Lack of a Safe Alternative
Opioids (including prescription opioid pain relievers and heroin) killed more than 28,000 people in the United States alone in 2014, more than any year on record[1]. At least half of all opioid overdose deaths involve a prescription opioid.
Outside of the US, such as in the UK, you can go into the Walgreens/Boots pharmacy, buy a £19.99 ($28.20) medical device, and get a significant reduction in pain and drug usage. Most interestingly, the 40% of the users are taking opioids and 87% have a reduction including 23% eliminating the drug use.
Chronic pain results from central sensitization of the nervous system, which causes a person to develop a persistent state of high reactivity to pain provocation and sensitivity. This also causes the amplified pain not to be correlated to the injured tissue or terminated after the injury has healed.
Pain Management published our 5,000+ survey results A UK registry study of the effectiveness of a new over-the-counter chronic pain therapy, Pain Manag. 2015 Nov; 5(6): 413-23 (http://www.futuremedicine.com/doi/full/10.2217/PMT.15.35) reported 2/3 of participants had more than 57% pain relief.
A follow up observational study was conducted to measure the medical device’s pain relief durability, its reduction of pain medication use, including prescription narcotic drugs, and its impact on the user’s quality of life.
Out of the 254 chronic pain sufferers that participated, 224 completed the full 6-month study.
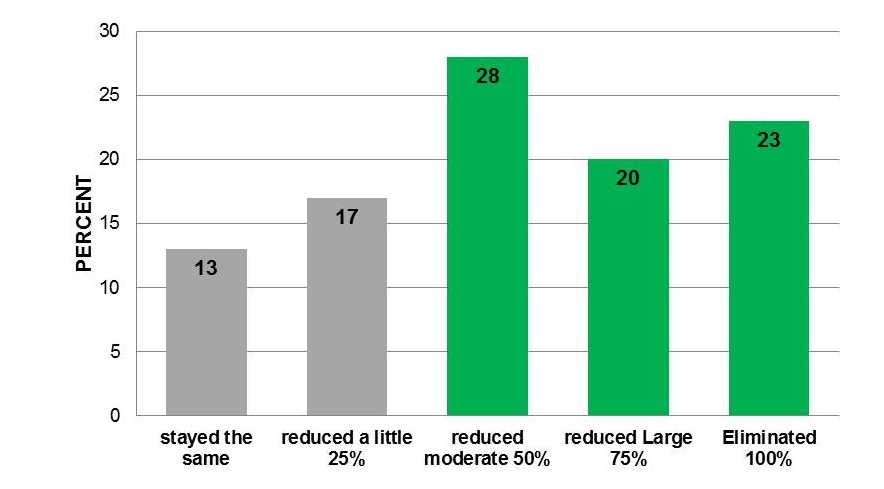
Opioid Medication Use 87% reduced use with 23 % having eliminated. Average decrease was greater than 50%.
The key findings:
- Side effects: None reported
- Treatment durability: Over 90% of the participants reported continued relief averaging a 61% reduction in pain even after six months.
- Improved quality of life: 90% reported improved sleep, 89% reported greater physical activity and 71% reported their quality of life was much better to a great deal better.
- Decreased medication use: 86% reported a decrease in medication use that continued to improve over time. Most importantly, this decrease included prescription narcotic based drugs.
- Decreased adverse events from medication use: 68% were experiencing adverse side effects, of these 71% reported a reduction in adverse side effects.
- Healthcare savings: 65% spent less on pain therapies and 65% reported fewer visits to the Doctor and Pain Specialist.
BioElectronics a small US company has been stalled in the US market by the US FDA’s years of regulatory reclassifications. The Company has on file since August last year another 510(k), K152432, for market clearance for the adjunctive use in the treatment of musculoskeletal pain that requires your leadership to provide physicians and consumers an affordable over-the-counter drug-free alternative to the devastating effects of drug therapy.
This safe and effective technology has been stalled in the US FDA’s years of regulatory reclassifications.
How you can help the opioid epidemic
We ask that you contact the new FDA Commissioner, Robert M. Califf, MD, to address 510(k) file number K152432 to expedite getting this drug-free relief technology to chronic pain sufferers:
- Email: Robert.Califf@fda.hhs.gov
- Mailing Address: Food and Drug Administration, 10903 New Hampshire Avenue, Silver Spring, MD 20093