August 4, 2014
Dear BIEL Shareholders:
BioElectronics Corporation’s mission is to change the way people heal and relieve their pain. As the chart below, that clearly demonstrates that, we have done exactly that.
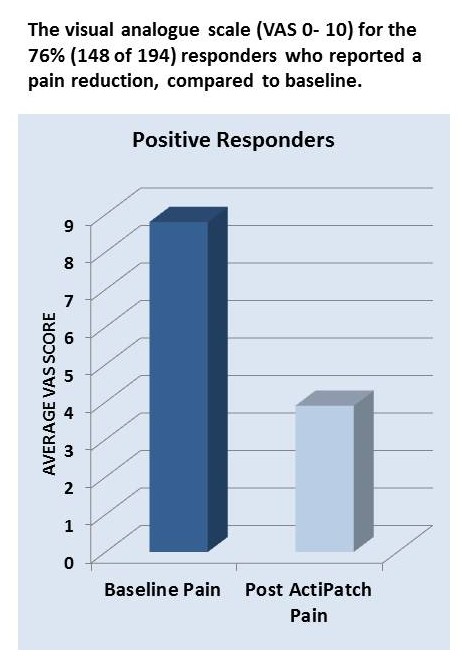 Chronic pain sufferers in the United Kingdom said that ActiPatch reduced their pain by more than 50%. This is what the BioElectronics team has worked so hard to achieve. It is also the reason so many of you have stuck with us through some terrible economic times and the frustration of the U.S. regulatory environment.
Chronic pain sufferers in the United Kingdom said that ActiPatch reduced their pain by more than 50%. This is what the BioElectronics team has worked so hard to achieve. It is also the reason so many of you have stuck with us through some terrible economic times and the frustration of the U.S. regulatory environment.
This year has been an exciting period of positive change for the medical device industry and especially for BioElectronics. I am anxious to share with you an update on the progress we made on the four-part growth strategy we initiated last year. Some high points were: our first successful retail launch at Boots in the UK; expanded distribution in Europe; successful clinical trials completed on osteoarthritis of the knees, long-term plantar fasciitis and lower back pain; and more awards from industry publications.
Central to our success in executing this strategy has been the addition of Dr. Deepak Kotak, M.D. to our senior leadership team. His multi-faceted strengths as a critical care physician, anesthesiologist and also as civil attorney in the UK, has been and continues to be invaluable. He has taken the lead in critical discussions with prospective partners for sales, distribution agreements, and technology licenses. His understanding of the many unmet medical needs, combined with a creative vision of BioElectronics patented technology, has added diversity and depth to the pipeline that will enable BioElectronics to lead and dominate the market. I am confident his leadership will serve shareholders for many years to come. Moreover, his current location in London has been critical in strengthening our engagement and visibility with European customers and partners.
We have made progress because we’ve stayed focused on the four strategic initiatives and expanding the scope as opportunities arose. Those ongoing efforts are:
- Executing an aggressive marketing and sales program in the large European market and other select global markets;
- Developing political and public support to induce the US FDA to recognize our safe therapeutic solution to address the dire need for an alternative to the devastating side effects, complications and addictions of OTC and prescription analgesic drugs;
- Continuing clinical research to develop new products and substantiate our efficacy in new indications with large markets and significant unmet medical needs; and
- Lowering our cost of capital to finance expansion.
First – Global Marketing Program
Sales of our ActiPatch® Therapy products are steadily increasing in Boots, the dominant pharmacy chain in the United Kingdom and a role model in the industry. The introduction last fall got a slow start because it was off-cycle from the Boots normal product introduction calendar and our product was initially only available in some Boots retail stores. Nevertheless, we have seen steady and now accelerating growth in sales, both online, and in the stores thanks in large measure to our social media sampling program, “Try It and Tell Us,” on Facebook.
This program has multiple advantages: it lowers the perceived risk of trying our innovative product by putting 7-day sample devices directly in the hands of the consumers poorly served by current analgesics; it supports word-of-mouth about the product’s efficacy; it is building a strong body of anecdotal testimonials and feeding our growing body of data on customer experience. It is especially promising that survey results indicate about 70% of those people with chronic pain, who have tried a sample, are planning to buy the product. This far exceeds the industry standard. You can read the first-hand reports at http://www.bielcorp.com/ or on the ActiPatch Facebook page.
The successful sales growth experience at Boots will facilitate and accelerate getting the product in more retail outlets around the world. Boots pharmacies are a strategic member of the Boots-Alliance, the world’s first global pharmacy-led health and well-being enterprise that includes Walgreens, the largest drugstore chain in the US.
Other global recognition for ActiPatch Therapy included citations in two award categories from the OTC Bulletin, the leading global trade publication for over-the-counter healthcare products. The Smart Insole™ Heel Pain Therapy product was highly commended as the “Most Innovative New OTC Product Award” and the ActiPatch® Musculoskeletal Therapy was runner-up for its “Best New OTC Packaging Design Award.”
We also expanded our geographic reach this year by adding new distribution partners with strong, medical distribution experience and existing product lines.
Second – Developing political and public support to induce the US FDA to recognize our therapeutic solution
This year, we saw some progress with the FDA. We were very pleased by the FDA recommendations announced in February in their Docket No. FDA-2012-N-0378, “Physical Medicine Devices; Reclassification and Renaming of Shortwave Diathermy for All Other Uses.”
By reclassifying this technology to reflect its safety and efficacy, the FDA appears to be making scientific evidence the determinant for additional market clearances, not out-dated categorizations. Once the agency finally makes a ruling (comments closed on May 21 and as of this date, we have no further news), the Company plans to submit new 510K applications with our extensive clinical data coming from the world’s leading medical institutions.
U.S. pain sufferers need ready access to this safe, drug-free and cost-effective technology that offers superior pain relief and accelerates healing. It will be twelve years next week since the FDA granted market clearance to us for the reduction of edema following blepharoplasty. Out-dated classifications put this breakthrough technology in a bureaucratic maze that delayed getting new indications and over-the-counter approval. Our ActiPatch Therapy is an over-the-counter electronic bandage that could give quick relief to millions of American pain sufferers at breakthrough prices.
Now with the new FDA approach to classification, we are hopeful that we will get equivalent U.S. market clearances to those we have earned in Canada, the European Common Market and much of the rest of the world at a faster pace. However, I want to assure shareholders that while the U.S. market is important, it is not critical to the success of BioElectronics. Our retail launch in the UK and the signing of new distributors around the world shows that BioElectronics Corporation will succeed with or without an FDA clearance for OTC in the U.S.
Third – Continuing high quality clinical research, to further substantiate clinical efficacy and establish more indications of use.
Since my last shareholder report, we have successfully completed three clinical studies, some observational studies, and collected a growing body of consumer testimonials and pain data. The double-blind placebo controlled studies were for the following indications: osteoarthritis of the knee, long-term plantar fasciitis and lower back pain.
The randomized, double blind, placebo controlled trial on osteoarthritis of the knee (knee pain) using ActiPatch® Therapy was conducted by Dr. Gianluca Bagnato, Division of Rheumatology, University Hospital Gaetano Martino, Messina, Italy. Results showed statistically significant improvements in all three measures for the group wearing the active ActiPatch device relative to the group wearing the non-active device.
At the Division of Sports Medicine, University of British Columbia, Dr. Jack Taunton completed a randomized, placebo-controlled crossover trial on chronic plantar fasciitis (heel pain) using ActiPatch Therapy. The study results showed strong improvement on a week-by-week basis, statistically above baseline at 4 weeks, 9 weeks and 26 weeks, with 26 weeks showing the most improvement for both pain and disability. Improvement at 26 weeks was the study primary end point and is indicative of tissue remodeling and healing.
An observational study conducted in Italy demonstrated the value of our technology in the large dental implant market. Dr. Bartolomeo Operti MD, Chief of Anesthesia Department Day Hospital Section of Maria Adelaide Hospital and Dr. Tiziano Tealdo, Clinical Instructor at University of Genova, Valle Belbo Implant Center have completed a white paper detailing their results regarding the use of BioElectronics medical device, RecoveryRx Therapy in treating swelling and pain in their dental implant patients following extractions.
Global leaders in research are continuing work on additional clinical studies at the following institutions: Tufts Dental School, University of British Columbia, University Hospital Ghent, Belgium, and University Hospital G. Martin, Messina, Italy.
Fourth — Lowering our cost of capital to finance expansion.
There are a number of potential applications of our underlying technology that we have had to put on the back burner because of resource issues. One way we are moving to reduce our cost of capital is by exploring the possibility of partnering in specific key markets with companies that have established sales and marketing presence.
We were also successful in getting the Depository Trust Company to end the deposit transaction restriction (the Deposit Chill) it had previously placed on shares of BioElectronics.
I will continue to keep you posted on the progress of our four-pronged strategy. The successful retail introduction in Boots is sparking the interest of other leading retailers around the world. We will continue to pursue FDA approval as new classification unlocks opportunity. We have a pipeline of new clinical studies that will support additional indications and new market opportunities. All of this will allow us to lower our cost of capital and achieve our potential.
Our core advantage continues to be the strength of our patented Bioelectroceutical technology that powers our current product line and is enabling a very strong growing pipeline of new products. The products in development will allow us to address additional lucrative markets in the years to come.
On behalf of the board and management of BioElectronics, I want to thank all our long-term investors for their patience and faith in the future of our Company. We will continue to expend every effort to fulfill its promise.
Sincerely,
Andrew J. Whelan
President
* * * * *
Contact: Paul Knopick, 940.262.3584, pknopick@eandecommunications.com
0 Comments Leave a comment